Pharma companies are in scope.
“Terrascope made it easy for our team to understand our emissions footprint and how we stacked up against industry peers. Their insights helped us focus on what really matters—driving reductions that align with both our sustainability goals and our bottom line..”
Paul Smith
Chief Operations Officer,
iNova Pharmaceuticals
4-6 Weeks
vs. 6-12 months traditional typical FLAG submission time frame.
100% Pass Rate
for audit and SBTi Approvals
600M+
tonnes of CO₂ measured across customers
92%
Accuracy vs. actual supplier data when estimating
200K+ Emission Factors
with FLAG, LUC, & LM breakdowns
80%
reduction in measurement cycles
50x Faster
than traditional LCAs for product emissions
5x Faster
faster measurement and savings of up to USD160k
10 years
of sustainability experience Terrascope is an Olam venture.
| Before Terrascope | After Terrascope | |
|---|---|---|
| Supplier coverage | Top 20 only | 80%+ of spend |
| API manufacturing data | Estimated | Supplier-specific |
| Cold chain visibility | Logistics only | Full temperature control impact |
| Packaging emissions | Ignored | Material and design level |
| Audit defensibility | Uncertain | Regulatory-grade trail |
Healthcare customers are asking for carbon data you don't have
- API manufacturing is energy-intensive but data is scarce
- Cold chain logistics add complexity most platforms can't handle
- Packaging requirements conflict with sustainability goals
- Healthcare procurement increasingly includes carbon criteria
WEasily meet audit and reporting demands with a defensible baseline
- Chemical and manufacturing emission factors for API production
- Cold chain and logistics carbon calculation
- Packaging lifecycle assessment capabilities
- Supplier engagement that works for regulated industries
- Audit trail that satisfies pharma compliance requirements
Turn hotspots into prioritized, investable decarbonization plans, all in one platform
- 200k+ emission factors including chemical manufacturing
- 92% accuracy vs actual supplier data
- Full audit trail for regulatory scrutiny
- IDC MarketScape Leader in APEJ Carbon Management
Tailored solutions for different company profiles
If you Have Data,
But Need Compliance
"We measured Scope 3 with industry averages or third-party tools. Now LSR regulations require land-specific breakdowns we don't have."
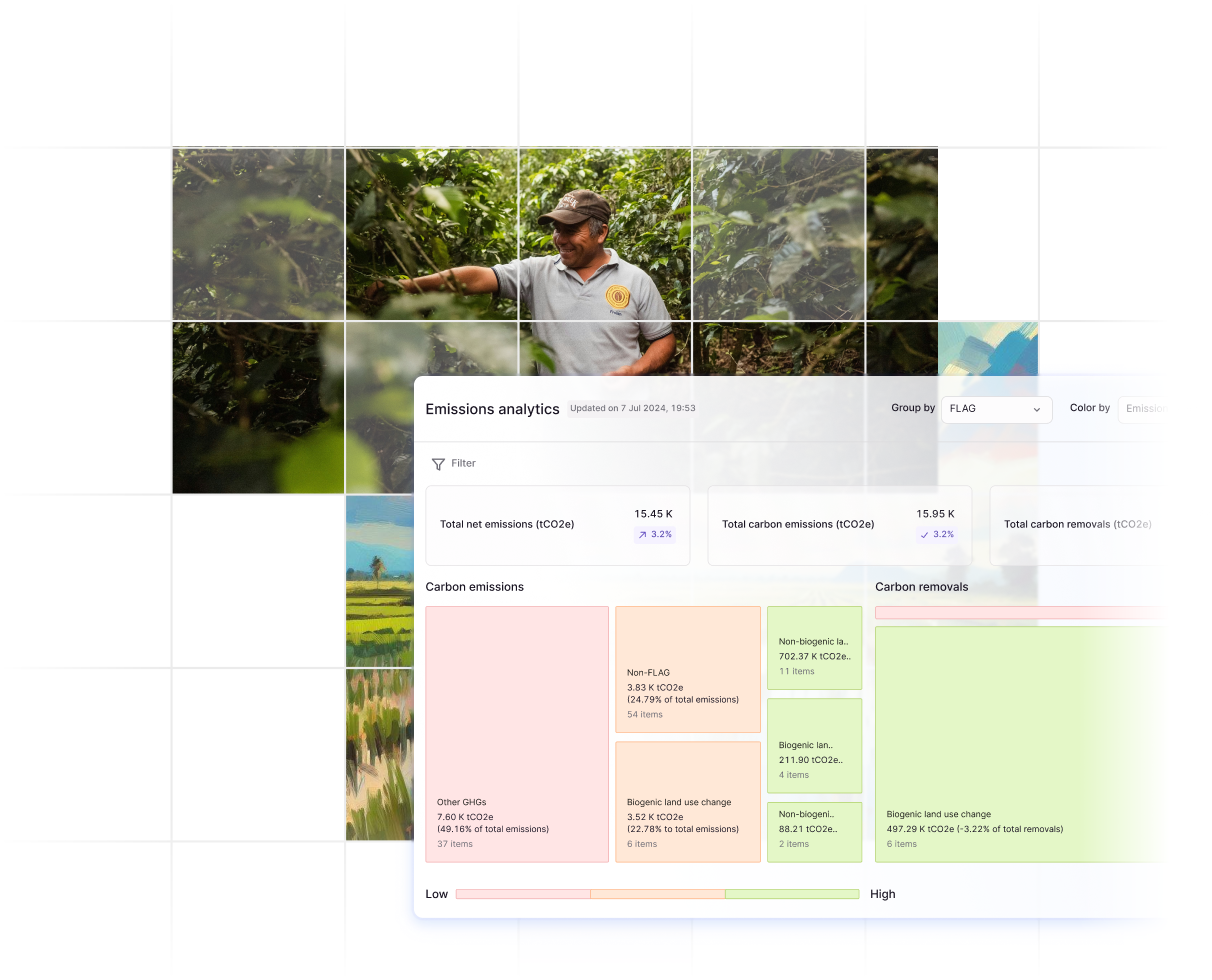
Land Sector EF Disaggregation
- Auto-tagging of proprietary emissions factor databases into LSR-specific requirements
- Monthly office hours with LSR Expert
Facing Customer/SBTi Pressure
"Customers want product-level carbon footprints. broken out by land emissions categories. Our industry-average numbers make us look worse than competitotrs with farm-level data."
"We need to quantify progress vs our SBTi targets."
Farm-level emissions calculation to prove lower impact and win contracts
- Proprietary, commodity-specific templates for farm-level data collection
- Scalable across owned farms and suppliers
- Monthly expert office hours included
- Monthly office hours with LSR Expert
Real Results from Industry Leaders
16% Lower Emissions vs Industry Benchmark Custom EF development
130K line items in 2 weeks at 92% accuracy
Measured 5,000 SKUs in 100 minutes
16% Lower Emissions vs Industry Benchmark Custom EF development
130K line items in 2 weeks at 92% accuracy
Measured 5,000 SKUs in 100 minutes
Regulatory Requirements for Pharmaceutical Companies
See which regulation applies to your jurisdiction, and what it means for your business.
Europe
| Regulation | Deadline | What It Means |
|---|---|---|
| CSRD | 2025-2028 | Large pharma companies in scope. Supply chain disclosure required. |
| EU Pharma Strategy | Ongoing | Environmental assessment in medicine lifecycle increasingly required. |
United States
| Regulation | Deadline | What It Means |
|---|---|---|
| California SB 253 | Aug 2026 | Major pharma companies with $1B+ revenue in scope. |
| Healthcare GPO Requirements | Ongoing | Group purchasing organizations adding carbon to supplier scorecards. |
Australia
| Regulation | Deadline | What It Means |
|---|---|---|
| AASB S2 | Jan 2025+ | Pharma companies with Australian presence. Supply chain scrutiny increasing. |
Singapore/APAC
| Regulation | Deadline | What It Means |
|---|---|---|
| SGX Requirements | FY2025 | Listed pharma companies need climate disclosure. |
FAQ
What if our data is incomplete (e.g., missing BOM details)?
It is common for pharmaceutical companies to have incomplete data, often around 70% coverage for Bill of Materials. Terrascope addresses this using AI-powered Emission Factor (EF) matching across multiple databases (such as Ecoinvent and ADEME) and driver trees to improve accuracy when specific process data is missing. This allows you to establish a baseline immediately while prioritizing primary data collection for high-impact hotspots.
How does the platform help with packaging compliance (PPWR) and EPR fees?
Terrascope provides specific guidance on evolving regulations like the EU Packaging and Packaging Waste Regulation (PPWR), helping clarify jurisdiction-specific definitions such as single-use versus reusable packaging. Furthermore, the platform helps model scenarios to reduce Extended Producer Responsibility (EPR) fees.
How do you handle Scope 2 complexities and audit requirements?
Terrascope solves for grid emission factor variances between manufacturing sites (e.g., accurately reflecting higher intensity grids like South Africa vs. Australia). Crucially for investors, the platform separates location-based and market-based Scope 2 reporting and provides transparent handling of Renewable Energy Certificates (RECs). This ensures your reporting is audit-ready for stakeholders who demand credible, comparable data.
Resources